 By Hannah Kopinsky, MD
By Hannah Kopinsky, MD
Peer Reviewed
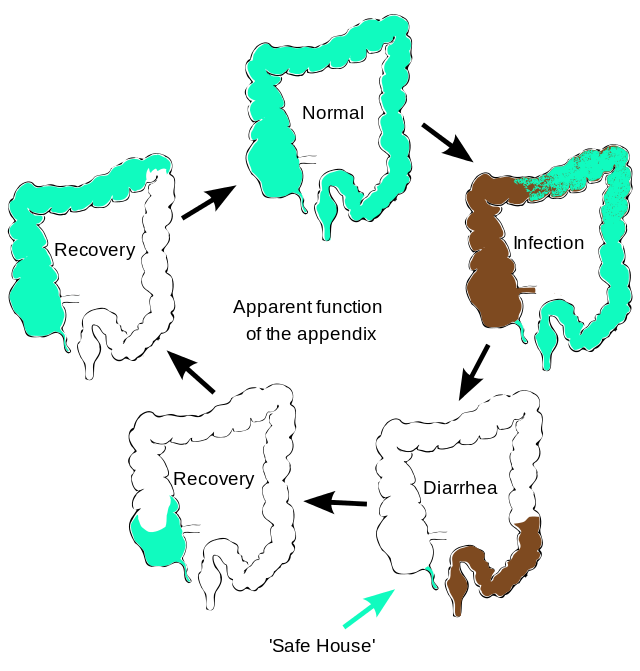
Appendicitis is the most common reason for urgent surgery related to abdominal pain in the US, with a lifetime incidence of 8.6% for men and 6.7% for women.1 The …

 By Hannah Kopinsky, MD
By Hannah Kopinsky, MD
Peer Reviewed
Appendicitis is the most common reason for urgent surgery related to abdominal pain in the US, with a lifetime incidence of 8.6% for men and 6.7% for women.1 The …

Clinical Correlations is proud to present our section for humanism in medicine, Tales from the Bedside, where we showcase our community’s unique and diverse experiences …