Jeffrey Mayne MD
Faculty peer reviewed
Dr. Okonkwo’s post this past summer to PrimeCuts summarized ongoing research in Alzheimer’s Disease (AD), the most common cause of dementia affecting 15 million individuals worldwide and more than 5 million in the United States. It is a disease marked by cognitive deterioration that slowly breaks down one’s ability to perform activities of daily living and leaves individuals unable to care for themselves.
Briefly, the diagnosis of AD is based on clinical criteria and histologic confirmation at death. Based on these findings, the disease is broken into three categories: definite, probable, and possible. Classic clinical features include an amnestic type memory impairment (a partial or total loss of antegrade memory and impaired learning with preserved general cognition), deterioration of language, and visuospatial deficits. Definitive diagnosis is by pathologic examination, though biopsy is not routinely performed because of the danger of the procedure, the cost, and low yield. In a patient who presents with symptoms suspicious for Alzheimer’s, brain imaging, preferably MRI, is performed.
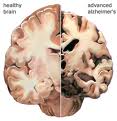
Imaging techniques differentiate AD from other causes of dementia, including cerebrovascular disease, structural diseases (i.e. chronic subdural hematoma, cerebral neoplasm, normal pressure hydrocephalus) as well as regional brain atrophy such as frontotemporal dementia. In AD there is nonspecific focal and diffuse atrophy, as well as white matter lesions, though some investigators have associated reduced hippocampal volumes with the disease. There are no laboratory tests to diagnose AD.
The Neurological and Communicative Disorders and Stroke and the Alzheimer’s Disease and Related Disorders Association (NINCDS-ADRDA) defines the clinical criteria for diagnosis as: (1) a standardized brief mental status exam; (2) deficits in two or more areas of cognition; (3) progressive worsening of memory; (4) no disturbance of consciousness; and (5) onset between ages 40 and 90. The pathological hallmarks include neuronal intracellular neurofibrillary tangles of hyperphosphorylated tau proteins and extracellular deposits of synaptotoxic ß-amyloid (Aß) peptides in fibril structures.
Current research in AD is focused on identifying the onset of the pathological processes, to detect AD in its earliest stages in order to gain a better understanding of its pathogenesis. The July 2009 JAMA article Use of Alzheimer Disease Biomarkers reports on the utility of cerebrospinal fluid (CSF) markers ß-amyloid (Aß42), total tau protein (T-tau), and tau phosphorylated at position threonine 181 (P-tau) for predicting incipient AD in patients with mild cognitive impairment (MCI). The research concludes that CSF Aß42, T-tau, and P-tau identify incipient AD with good accuracy, that these biomarkers could be used to predict outcomes, and that they could potentially be useful in identifying patients for clinical trials and for screening tests in memory clinics.
Of critical importance is the fact that at this time no treatment exists to prevent or alter the course of the disease. A recent review by the American College of Physicians and the American Academy of Family Physicians found little evidence that drug treatments are clinically meaningful. Three of the most common medications used in practice today are donepezil, rivastigmine and galantamine. These work by reversibly and noncompetitively inhibiting centrally active acetylcholinesterase, the enzyme that hydrolyzes acetylcholine, resulting in increased concentrations of acetylcholine available for synaptic transmission in the central nervous system. Galantamine also modulates the nicotinic acetylcholine receptor to increase acetylcholine from surviving presynaptic nerve terminals.
Current FDA approved treatment for AD consists of the cholinesterase inhibitors, including donepezil. Overall, cholinesterase inhibitors such as tacrine, donepezil, and rivastigmine have shown marginal benefits, though their effects have been shown to be statistically significant. Importantly, they are generally well tolerated by patients; the adverse effects on the alimentary tract associated with the cholinesterase inhibitors are transient and occur in a small percentage of cases. Other medications either in clinical trials or for which only preliminary data exists include the antioxidant vitamin E, drugs such as selegine and memantine (an NMDA antagonist), anti-inflammatory agents (such as prednisone, diclofenac, rofecoxib, and naproxen), as well as hormone-replacement therapies. A trial by Aisen, Scheider, Sano, et al. reported in JAMA determined that treatment with high-dose vitamin B supplements for 18 months did not slow cognitive decline in individuals with mild to moderate AD.
Ongoing research is aimed at identifying AD in its earliest stages and preventing its progression. Chelators, which may help in promoting the degradation of amyloid beta plaques have been studied in phase II randomized controlled trials, with treatment associated with a dose-dependent reduction in amyloid beta-42 levels in the cerebrospinal fluid. In other research the inhibition of beta-secretase has been shown to have a dose-related effect on activities of daily living and global function.
Researchers are also investigating the possibility of vaccination. Early studies in mice have shown that immunization with amyloid beta peptide reduced amyloid cerebral angiopathy (the accumulation ofAß in the wall of vascular vessels). A study published in the Lancet in July 2008 showed a decrease in post-mortem amyloid cerebral angiopathy in eight immunized patients compared to non-immunized controls. Despite the results, seven of eight patients had severe end-stage dementia at the time of death. Additionally,, immunized patients had a greater amyloid beta plaque burden in cortical blood vessels compared to non-immunized patients. Although in this study, vaccinated patients experienced a progression of disease to the final stage, there are a number of studies indicating beneficial effects of the treatment on the disease course, even though these trials were prematurely halted.
Finally, the use of CSF biomarkers is being explored, with the hope of identifying the disease in its earliest stages before the cognitive effects are apparent. Future research will utilize neuroimaging techniques, including positron emission tomography (PET), FDG-PET or SPECT scans, which reveal distinct regions of low metabolism and hypoperfusion in AD. In addition. PiB (Pittsburg Compound B) PET will be integrated with biomarker data to identify the optimal panel to use for predictive testing for AD, AD diagnosis and clinical trial monitoring.
Dr. Mayne is a 1st year resident in internal medicine at NYU Medical Center.
References:
1.) Blennow K, deLeon MJ, Zetterberg H. Alzheimer’s disease. Lancet. 2006;368(9533):387-403.
2.) Cummings J. Alzheimer’s Disease. N Engl J Med. 2004; 351:56-67.
3.) Knopman, DS, DeKosky, ST, Cummings, JL, et al. Practice parameter: diagnosis of dementia (an evidence-based review). Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2001; 56:1143.
4.) Silverman, DH, Small, GW, Chang, CY, et al. Positron emission tomography in evaluation of dementia: Regional brain metabolism and long-term outcome. JAMA 2001; 286:2120.
5.) McKhann G, Drachman D, Folstein M, et al. Clinical Diagnosis of Alzheimer’s disease: report of the NINCDS-ADRDA Work Group under the auspices of the Department of Health ad Human Services Task Force on Alzheimer’s Disease. Neurology 1984;34:939.
6.) Mattsson N, Zetterberg H, Hansson O, et al. CSF biomarkers and incipient Alzheimer disease in patients with mild cognitive impairment. JAMA. 2009;302(4):385-393.
7.) Savva G, Wharton S, Forster G, et al. Age, Neuropathy, and Dementia. N Eng J Med. 2009;360(22)2302-2309.
8.) Voelker R. Guideline: dementia drugs’ benefit uncertain. JAMA. 2008;299(15):1763.
9.) Aisen PS, Scheider LS, Sano M, et al. High-dose vitamin B supplementation and cognitive decline in Alzheimer disease. JAMA. 2008;300(15):1774-1783.
10.) Friedrich MJ. Therapeutic Environmental Design Aims to Help Patients with Alzheimer Disease. JAMA. 2009;301(23):2430-2433.
11.) Holmes C, Boche D, Wilkinson D, et al. Long-term effects of Abeta42 immunisation in Alzheimer’s disease: follow-up of a randomized, placebo-controlled phase I trial. Lancet. 2008; 372(9634):216-23.

One comment on “Alzheimer’s Disease 2.0”
Comments are closed.