Commentary by Jamie Hoffman, MDÂ
A healthy 54 year old man without past medical history presents for a routine physical exam for his insurance company. His blood work reveals a total protein (TP) of 9.4 g/dl and an albumin of 3.0 g/dl. What should be included in this patient’s diagnostic workup?
An elevated TP:Albumin ratio often necessitates finding the protein(s) responsible for such an elevation. Plasma proteins largely consist of albumin and globulins such as immunoglobulins, carrier proteins, and acute phase reactants. Elevated globulin levels are concerning. An important question to ask oneself in the workup of elevated proteins is whether there is an increase in multiple immunoglobulins (i.e. polyclonal gammopathy like HIV, viral hepatitis, liver disease, connective tissue disease or anything that stimulates a generalized immune response) or in one specific ‘clone’ (i.e. monoclonal, produced by a malignant plasma cell or other B-cell malignancy).
Monoclonal proteins are made by the proliferation of a single clone of plasma cells. In order to assess for the presence of a monoclonal protein, an SPEP should be ordered. In this test, a patient’s serum is placed into a agarose medium that separates the proteins based on size and charge (+ charge on left).Â
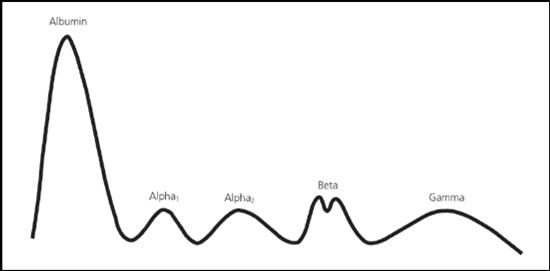 For reference, a normal SPEP:Â
For reference, a normal SPEP:Â
Alpha-1 fraction= alpha-1 antitrypsin, thyroid binding globulin.
Alpha-2 fraction= ceruloplasm, haptoglobin.
Beta-1= tranferrin Beta-2= beta-lipoprotein [IgA, IgM, even IgG at times].
                                                                              Between Beta and Gamma= CRP, fibrinogen.
                                                                              Gamma= immunoglobulins. (1)
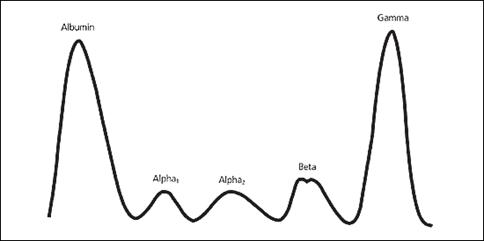
Typical Gamma M Spike meaning that we a have a large amount of a very specific protein (i.e. produced by a clone).
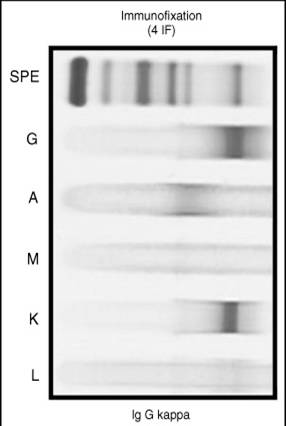
For further characterization of the M-spike, an immunofixation (IF) study should be ordered. IF uses monoclonal antibody probes to determine the isotype of a monoclonal protein (i.e. ‘IgG Kappa’- see IF below). IF is often used when the presence of an M-protein cannot be ascertained using SPEP, but clinical suspicion is high. A newer test known as serum free light chains is even more sensitive, and has rendered true ‘non-secretory myeloma’ to be a rare entity.(2)
To look for monoclonal protein in the urine, UPEP and urine IF can also be useful. Lastly, a hematologist may be needed for a bone marrow biopsy as well.
Note the discreet bands in the IgG lane and the Kappa lane- clearly identifying a kappa restricted (i.e. monoclonal) IgG protein.
Once the above diagnostic studies are obtained, the physician can often use clinical signs and symptoms along with biopsy results and M spike characteristics to help define the clinical disease state. For example:
• MM: clonal plasma cells in marrow (or plasmacytoma) AND end organ damage [CRAB- hyperCalcemia, Renal dysfxn, Anemia or lytic Bone lesions].
• MGUS: (if M-spike is ≤3g/dl and <10% plasma cells in bone marrow)
• Smoldering myeloma (SMM): (M-spike>3 g/dl or >10% plasma cells)
One should be mindful of the patient who presents with elevated proteins plus nephrotic syndrome, carpal tunnel syndrome, autonomic neuropathy and/or cardiomyopathy. Amyloidosis should be high on the differential as the plasma cells produce amyloid proteins that deposits in variable organs. A fat pad biopsy can help to cinch the diagnosis.
Patients with significant protein abnormalities should be referred to a hematologist for evaluation and treatment as a variety of options exist and many protein disorders carry serious risks and progression to more serious entities. Treatment options for MM, while largely incurable, include conventional agents like melphalan, steroids, or auto-transplant as well as newer agents like thalidomide, lenalidomide or Bortezomib (Velcade). The risk of progression to MM from MGUS is 1-2% per year and patients need to be assessed yearly for signs of CRAB. For SMM, the risk is higher (10-15% per year), and follow-up is warranted every 3 to 4 months. An abnormal serum free light chain ratio (K/L ratio) is also predictive of progression to frank multiple myeloma (hazard ratio, 3.5; 95% confidence interval [CI], 2.3-5.5; P < .001). This progression is independent of the size and type of serum monoclonal (M) protein. (3)
1. Â O’connell, T. “Understanding and Interpreting Serum Protein Electrophoresis”. Am Fam Phys 2005; 71:105-12.
2. Â Drayson M et al. “Serum free light-chain measurements for identifying and monitoring patients with nonsecretory multiple myeloma” Blood, 1 May 2001, Vol. 97, No. 9, pp. 2900-2902.
2. Rajkumar, V e t al. “Serum free light chain ratio is an independent risk factor for progression in monoclonal gammopathy of undetermined significance”. Â Blood, 1 August 2005, Vol. 106, No. 3, pp. 812-817.

3 comments on “Elevated Total Protein and the Interpretation of Serum Protein Electrophoresis”
what is the significance of a borderline Beta Globulin, normal LDH, normal CRP and ESR. Patient has generalized myalgia, worse in upper body and wrists. Is obese, other labs ok. any assist would be appreciated.
Comments are closed.