Peer Reviewed
This week in the news, House Republicans at long last unveiled the American Health Care Act, their replacement plan for the ACA. Meanwhile the Justice Department decided to clean house by asking for the immediate resignation of all Obama-appointed district attorneys, with swift consequences for those who did not comply. President Trump debuted version 2.0 of his executive order regarding immigration and refugee status from 6 Muslim majority nations. South Korea, four years after electing its first female president, saw another important first with her impeachment over an ongoing corruption scandal. All this upheaval is enough to give anyone palpitations, but luckily in this week’s Primecuts we can feel more assured sending our palpitation prone patients into strong magnetic fields (no comment on the safety of watching cable news). Â
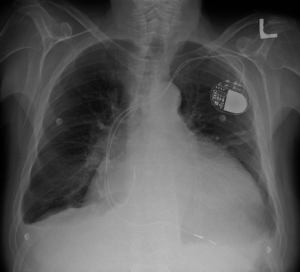
Assessing the Risks Associated with MRI in Patients with a Pacemaker or Defibrillator1
This study seeks to establish guidelines for the safe use of MRI in patients with implantable cardiac devices. Pacemakers or ICDs are generally a contraindication to MRI imaging due to concerns that strong magnetic fields used in MRI can cause heating of implanted devices, leading to myocardial injury. There are FDA approved MRI-conditional devices that have been shown to pose no known risk to patients under certain specified conditions. However, an estimated 2 million people in the United States have non-MRI-conditional implanted devices, potentially excluding them from a vital and versatile imaging modality.
In this study, adults with a non-MRI-conditional pacemaker or ICD who had a clinical indication for a non-thoracic MRI, underwent MRI after reprograming of their device (including no-pacing mode or inactivation of bradycardia or tachycardia therapies) Â , and were under careful observation to assess for any adverse events. The primary endpoints of the study were immediate consequences of MRI, including death, arrhythmia, or device malfunction requiring immediate replacement or repair. Secondary endpoints looked at changes in the device settings over several months.
The results of 1246 patients who underwent 1500 MRIs showed no deaths or device failures in patients who underwent imaging with appropriately reprogrammed devices. There was 1 patient who required immediate ICD replacement, however his device had not been appropriately reprogrammed according to the study protocol. Additionally, there were no episodes of ventricular arrhythmia and 6 episodes of atrial arrhythmias that spontaneously resolved within 49 hours.
One important limitation of this study is that it included only non-thoracic MRIs at 1.5 Tesla (which is the standard strength of the magnetic field used by most commercial MRIs). It is unclear what, if any, differences would be seen if the imaging field was directly over the implanted device or if the stronger magnetic field of newer 3T (3 Tesla) MRIs had been used. Additionally, patients with pacing dependent ICDs were excluded due to difficulty in appropriately reprogramming such devices prior to imaging. However, despite these limitations this study shows that in appropriately screened patients, and with close device monitoring, a previously avoided imaging modality can safely be used in patients with implanted cardiac devices.
Breast Cancer Screening in Denmark: A Cohort Study of Tumor Size and Overdiagnosis2Â
This retrospective cohort study looks at the change in incidence of advanced breast cancer diagnosed in Danish women as a result of initiation of breast cancer screening. In Denmark, routine breast cancer screening for women aged 50 to 69 with biennial mammograms was initiated in select areas in the early 90s, providing a cohort comprising of 20% of Danish women (284,140 women in 2010) in the screened group, and the remaining 80% in the non-screened control group (1,136,560 women in 2010). Diagnoses of breast cancer amongst all Danish women aged 35 to 84 were collected from 2 well validated national registries. The authors then compared the incidence rates of breast cancer both before and after screening was initiated, as well as within the screened group and the control group.
The results showed that the initiation of routine screening for women aged 50 to 69 did not lead to a lower incidence of advanced tumors. However, they did find that the incidence of non-advanced tumors (defined as tumors less than 20mm) did increase after screening was initiated. The authors calculated the number of over diagnosed tumors as the difference between the number of tumors in the screening areas and the non-screening areas, and estimated that the rate of over diagnosis of invasive breast cancer was between 14.7% and 38.6% (excluding DCIS), depending on which model was used.
This study is unique in that it not only compares rates of breast cancer before and after screening, but also compares the incidence of breast cancer in women who did not undergo screening before and after screening was initiated. This helps to control for any population wide changes in incidence over the study period.
The results of this study indicate that a significant number of women may be negatively impacted by routine breast cancer screening if they undergo unnecessary testing and treatment of tumors that would never have evolved to cause clinical significance. Further research is needed to help clinicians differentiate between clinically significant and insignificant tumors, and further work is needed on the part of clinicians to educate our patients about the potential for false positive results with routine screening.Â
Intensive Speech and Language Therapy in patients with chronic aphasia after stroke: a randomized, open-label, blinded-endpoint, controlled trial in a health-care setting3Â
This multicenter, parallel group, randomized controlled trial attempts to demonstrate the efficacy of intensive language therapy in treating post-stoke chronic aphasia. The authors of this study specifically chose the chronic aphasia subgroup, defined as aphasia lasting 6 months or longer post stroke, to challenge previously held assumptions that after initial improvement in speech and language function, patients with chronic aphasia were unable to further benefit from intensive language treatment.
In this study patients aged 18-70, who had chronic aphasia secondary to either ischemic and hemorrhagic stroke and had the ability to follow simple instructions were randomized to two groups. The first group received 3 weeks of intensive (greater than 10 hours/week) speech and language therapy, while the second group had a 3-week deferral period, before also receiving 3 weeks of intensive therapy. Both groups were then followed for 6 months, during which they could continue to receive language therapy as covered by their insurer. The primary end point was change in verbal communication effectiveness in everyday life scenarios as determined by the ANELT-A scale from baseline. The authors showed that verbal communication significantly improved by a mean of 3 points from baseline following 3 weeks of intensive speech and language therapy, and that these gains were sustained at 6 months. A subgroup which received 5 weeks of intensive therapy showed a mean improvement of 4 points from baseline. In comparison, previous studies showing improvement in acute (3 weeks post stroke) and post-acute (3 months post stroke) aphasia, have shown ANELT-A improvements of 10 points and 5 points respectively.
This study is unique in that it demonstrates that patients with chronic aphasia are able to benefit from intensive language therapy to a smaller but still significant degree compared to gains seen by patients with acute aphasia, and shows that these gains can be sustained over time. This study also suggests that longer duration of therapy may lead to an even greater benefit as seen by the improvements seen in the subgroup that continued to receive therapy. As this study was conducted in a real-world environment, which included basing duration of therapy on insurance coverage, it provides direct data that intensive speech and language therapy should be covered by insurance for at least 3 weeks, but potentially for longer, to allow for optimal recovery of communication ability in post-stroke patients.
Blinatumomab versus Chemotherapy for Advanced Acute Lymphoblastic Leukemia4Â
This is a multi-institutional phase 3 randomized control trial studying the improved survival of patients with refractory B-cell ALL treated with blinatumomab vs standard chemotherapy. Blinatumomab is a bispecific antibody that binds to CD-19, a B cell surface antigen expressed by more than 90% of B-cell precursor ALL blasts and to CD3-positive cytotoxic T cells, allowing for T cell mediated destruction of B cells. Patients included in this study were aged 18 or older with refractory B-cell ALL, defined as either refractory to induction or salvage chemotherapy, or first relapse within 12 months of fist remission, or any subsequent relapse. Patients were randomized to receive either induction and consolidation treatments with Blinatumomab or further chemotherapy with a combination of agents.
The primary endpoint evaluated in this endpoint was overall survival. The results showed that median survival in the blinatumomab treated group was 7.7 months as measured from randomization compared to 4 months in the chemotherapy group, results that led to early termination of the study because of the clear benefit observed with blinatumomab therapy. Additionally, patients in the blinatumomab group had higher rates of remission and longer duration of remission compared to the chemotherapy group.
This study shows very promising results of improved survival in patients with refractory ALL, in which 3-5 year survival is generally under 10%. The authors suggest that the efficacy of blinatumomab may be even more robust if combined with immune activation, given all patients in this study had been exposed to immunosuppressive chemotherapy, potentially dampening its immune-based effects. It is also possible to see enhanced effects of blinatumomab in combination with therapies that target additional B-cell receptors, such as rituximab and inotuzumab.
Despite these promising advances, it is important to note that at $178,000 per treatment5, blinatumomab is one of the most expensive cancer treatment drugs. As access to health care coverage is surely to be hotly debated over the next several months, if not years, it will be interesting to note what regulations, if any, are placed to control the costs of targeted cancer treatments such as this, which provide significant benefit to a small number of people at an astronomical cost.Â
Mini-Cuts
Linking Immunizations Status and Eligibility for Welfare and Benefits Payments: The Australian “No Jab, No Pay” Legislations6
With increases in outbreaks of preventable diseases, public health policy makers must grapple with the balance between personal liberty and community health. Legislators in Australia have greatly improved the rates of childhood immunizations by predicating childhood immunization for welfare benefits, and provide a model for similar application in the US.
The Promise of a boy: Indian Women are being mis-sold drugs to change their babies’ sex7
Social, economic, and cultural preferences for a male child are putting significant pressure on women in India to take drugs marketed as sex-selecting, with a prevalence as high as 60% amongst women living in certain rural regions whose first child was female. This illegal market is thought to directly contribute to higher numbers of miscarriages as well as widening gender gaps.
Modeling Contagion Through Social Networks to Explain and Predict Gunshot Violence in Chicago, 2006 to 20148
Comparing all arrests in Chicago with all recorded episodes of gun violence during the same time period, researchers were able to create a social network of gun violence that is better able to predict future victims of gun violence based on prior social associations than current models that are more heavily based on demographic and geographic data. This model suggests that gun violence can be transmitted through social interaction in a manner similar to infectious disease.
Dr. Irina Dimitrova is a 1st year resident at NYU Langone Medical Center
Peer reviewed by David Kudlowitz, chief resident, NYU Langone Medical Center
Image courtesy of Wikimedia Commons
References:
- Russo, Robert J., et al. “Assessing the Risks Associated with MRI in Patients with a Pacemaker or Defibrillator.†New England Journal of Medicine, vol. 376, no. 8, 2017, pp. 755–764. http://www.nejm.org/doi/full/10.1056/NEJMoa1603265?af=R&rss=currentIssueCopyright&
- Jørgensen, Karsten Juhl, et al. “Breast Cancer Screening in DenmarkA Cohort Study of Tumor Size and OverdiagnosisBreast Cancer Screening in Denmark.†Annals of Internal Medicine, American College of Physicians, 7 Mar. 2017, annals.org/aim/article/2596394/breast-cancer-screening-denmark-cohort-study-tumor-size-overdiagnosis. Accessed 11 Mar. 2017. http://annals.org/aim/article/2596394/breast-cancer-screening-denmark-cohort-study-tumor-size-overdiagnosis
- Breitenstein, Caterina, et al. “Intensive Speech and Language Therapy in Patients with Chronic Aphasia after Stroke: a Randomised, Open-Label, Blinded-Endpoint, Controlled Trial in a Health-Care Setting.†The Lancet, 2017, doi:10.1016/s0140-6736(17)30067-3. https://www.ncbi.nlm.nih.gov/pubmed/28256356
- Kantarjian, Hagop, et al. “Blinatumomab versus Chemotherapy for Advanced Acute Lymphoblastic Leukemia.†New England Journal of Medicine, vol. 376, no. 9, Feb. 2017, pp. 836–847., doi:10.1056/nejmoa1609783. http://www.nejm.org/doi/full/10.1056/NEJMoa1609783?af=R&rss=currentIssue
- Pierson, Ransdell. “Exclusive: Amgen’s New Leukemia Drug to Carry $178,000 Price Tag.†Reuters, Thomson Reuters, 17 Dec. 2014, www.reuters.com/article/us-amgen-cancer-exclusive-idUSKBN0JV1YU20141217. Accessed 11 Mar. 2017. http://www.reuters.com/article/us-amgen-cancer-exclusive-idUSKBN0JV1YU20141217
- Yang YT, Studdert DM. Linking Immunization Status and Eligibility for Welfare and Benefits PaymentsThe Australian “No Jab, No Pay†Legislation. JAMA. 2017;317(8):803-804. doi:10.1001/jama.2017.0123 https://www.ncbi.nlm.nih.gov/pubmed/28245331
- Cousins , Sophie. “The Promise of a Boy: Indian Women Are Being Mis-Sold Drugs to Change Their Babies’ Sex.†BMJ, British Medical Journal Publishing Group, 6 Mar. 2017, doi.org/10.1136/bmj.j913. Accessed 11 Mar. 2017.
- Green B, Horel T, Papachristos AV. Modeling Contagion Through Social Networks to Explain and Predict Gunshot Violence in Chicago, 2006 to 2014. JAMA Intern Med. 2017;177(3):326-333. doi:10.1001/jamainternmed.2016.8245 http://jamanetwork.com/journals/jamainternalmedicine/fullarticle/2594804