Podcast: Play in new window | Download
Subscribe: RSS
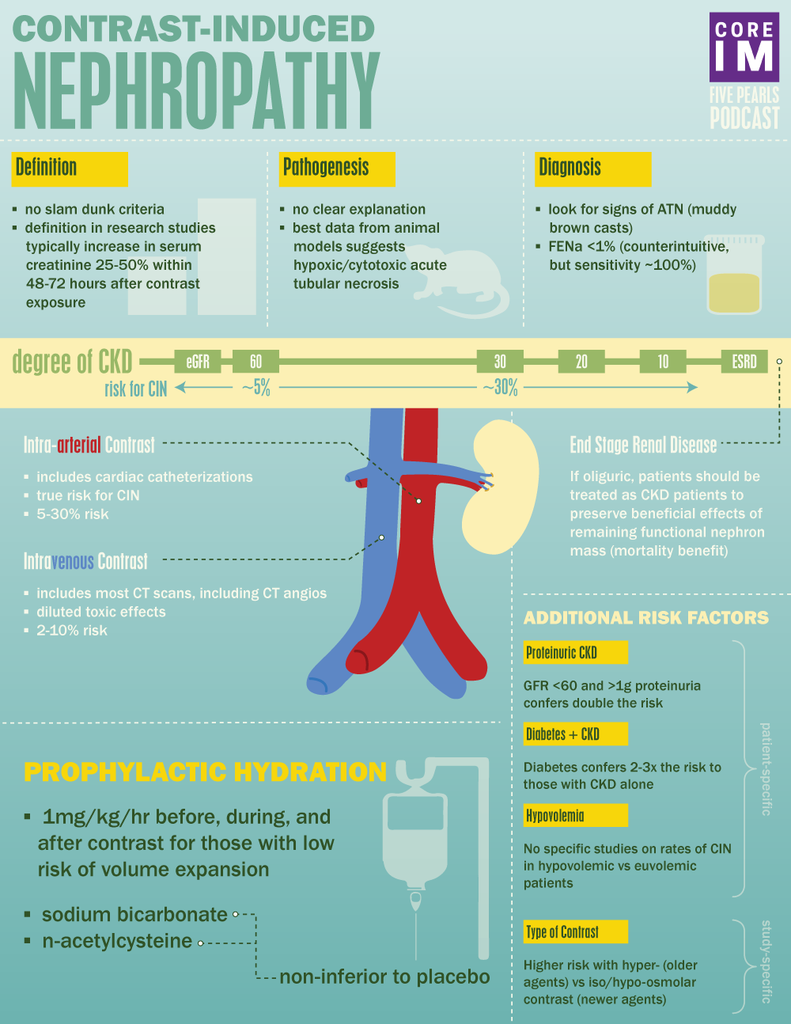 By Daniel Sartori MD, Marty Fried MD, Shreya Trivedi MD; Illustration by Michelle Lo MD and Amy Ou MD. Quiz yourself on the 5 Pearls we will be covering:
By Daniel Sartori MD, Marty Fried MD, Shreya Trivedi MD; Illustration by Michelle Lo MD and Amy Ou MD. Quiz yourself on the 5 Pearls we will be covering:
- Are there diagnostic criteria for contrast-induced nephropathy (CIN)? (2:11)
- Is there a difference between exposure to intra-arterial versus intra-venous contrast in terms of risk of CIN? (6:28)
- What are the biggest risk factors for CIN? (14:48)
- What preventive measures have been shown to best reduce the risk of CIN? (19:41)
- Can ESRD patients on hemodialysis still suffer from CIN? (23:32)
- Recap (25:49)
A special thank you to Dr. Josh Farkas for peer-reviewing this podcast!
Subscribe to CORE IM on any podcast app! Follow us on Facebook @Core IM || Twitter @COREIMpodcast || Instagram @core.im.podcast. Please give any feedback at COREIMpodcast@gmail.com.
Pearl 1:Â Diagnostic criteria for Contrast-Induced Nephropathy (CIN)
- Most research studies define CIN as a relative increase in serum creatinine of anywhere from 25-50% or, an absolute rise in serum creatinine of 0.5 to 1mg/dL within 48-72 hours after exposure to contrast with exclusion of other causes of acute kidney injury.
- Other clues are signs of acute tubular necrosis and a fractional excretion of sodium (FeNA) less than 1.
Pearl 2: Intravenous vs Intra-arterial contrast
- Most data suggest that CIN is a disease entity almost entirely reserved for patients undergoing intra-arterial contrast loads, and not intra-venous contrast loads.
- The incidence of AKI after arterial contrast ranges anywhere from 5-30% depending on the other risk factors present.
- The incidence of AKI following intravenous contrast exposure is much lower, in the ballpark of 2-10%
- Issues often with studies of arterial contrast:
- Retrospective
- No control group
- Selection Bias
- Other variables: showering atherosclerotic emboli to kidneys or volume of contrast used
- Issues often with studies of venous contrast:
- Also retrospective
- Controlled, but poorly
- Selection bias
Pearl 3: Risk Factors
- CKD
- Patients with underlying CKD are at increased risk of developing CIN
- Pinpointing the GFR at which the greatest risk occurs is very difficult because most studies lump patients into buckets with wide ranges of GFR
- What we do know:
- In patients undergoing studies with arterial contrast:
- ~ 5% of patients with GFR 30-60 develop CIN
- ~ 30% of patients with GFR <30 develop CIN
- Proteinuria
- GFR < 60 plus at least 1 g of proteinuria develop CIN at almost double the rate of patients with GFR <60 alone
- Diabetes:
- Patients with diabetes coupled with CKD demonstrated almost triple the rate of CIN compared to patients with CKD alone
- Hypovolemia:
- Given difficulty measuring volume status by exam or labs, there are no known randomized trials looking at rates of CIN in those hypovolemic vs. euvolemic
- But given data with prophylactic hydration, low effect arterial blood volume and hypoperfusion to the kidneys are thought to constitute elevated risk.
- High Osmolality Contrast
- Older contrast agents are hyper-osmolar compared to serum and associated with much higher rates of CIN
- Newer contrast agents are either iso-osmolar or hypo-osmolar and associated with rates that are lower relative to hyper-osmolar agents
Pearl 4: Prevention
- Isotonic fluid pre and post contrast study:
- Studies generally show that 24 hour of hydration with isotonic saline does in fact reduce risk of CIN compared to patients who did not receive it
- Scrutinizing the data:
- Could fluid administration just be preventing hypotension-induced AKI and not really represent a protective measure against contrast-induced AKI?
- Are these studies findings true for patients with high risk features (CKD, proteinuria etc)?
- Scrutinizing the data:
- Studies generally show that 24 hour of hydration with isotonic saline does in fact reduce risk of CIN compared to patients who did not receive it
- Sodium bicarbonate pre and post contrast study:
- Believed to work by alkalinizing the renal tubules and preventing free radical generation
- Sodium bicarbonate is thought to be non-inferior to normal saline for risk of developing CIN
- N-acetylcysteine (NAC)
- Rationale is that NAC reduces free radicals generation adding back a reducing agent
- Oral NAC demonstrated no reduction in CIN compared to placebo
Pearl 5: CIN in End Stage Renal Disease
- ESRD patients who are oliguric still have some functioning nephrons
- Residual kidney function has several beneficial effects
- Still allows for filtration of uremic toxins
- Still allows for some degree of volume and electrolyte regulation
- Most importantly, has survival benefit
- Patients with ESRD with some residual kidney function are thought to be at risk of CIN
- Currently, the American College of Radiology (ACR) describes this as a ‘theoretical risk:’ “This remains speculative, as there are no conclusive outcome data in oliguric dialysis patients in this setting.â€
References
Solomon R. Contrast-medium-induced acute renal failure. (1998). Kidney International, Vol. 53, pp. 230–242 .
Espinel CH, Gregory AW. (1980). Differential diagnosis of acute renal failure. Clin Nephrol;13(2):73.
Schwab SJ, Hlatky MA, Pieper KS, Davidson CJ, Morris KG, Skelton TN, Bashore TM. (1989). Contrast nephrotoxicity: a randomized controlled trial of a nonionic and an ionic radiographic contrast agent. N Engl J Med;320(3):149.
Farkas, Josh. “Do CT Scans Cause Contrast Nephropathy?†EMCrit. 29 March 2015, emcrit.org/pulmcrit/do-ct-scans-cause-contrast-nephropathy/.
(13:40) Ryan D. Aycock, MD, MS; Lauren M. Westafer, DO, MPH; Jennifer L. Boxen, MLS, MA; Nima Majlesi, DO; Elizabeth M. Schoenfeld, MD, MS; Raveendhara R. Bannuru, MD, PhD. (2018). Acute Kidney Injury After Computed Tomography: A Meta-analysis. Annals of Emergency Medicine. 71: (1). 44-53.
(14:03) McDonald JS, McDonald RJ, Carter RE, Katzberg RW, Kallmes DF, Williamson EE. (2014). Risk of intravenous contrast material-mediated acute kidney injury: a propensity score-matched study stratified by baseline-estimated glomerular filtration rate. Â Radiology. ;271(1):65. Epub 2014 Jan 16.
(14:03) McDonald RJ, McDonald JS, Bida JP, Carter RE, Fleming CJ, Misra S, Williamson EE, Kallmes DF. (2013). Intravenous contrast material-induced nephropathy: causal or coincident phenomenon? Radiology.;267(1):106. Epub 2013 Jan 29.
(16:59) Piskinpasa S, Altun B, Akoglu H, Yildirim T, Agbaht K, Yilmaz R, Peynircioglu B, Cil B, Aytemir K, Turgan C. An uninvestigated risk factor for contrast-induced nephropathy in chronic kidney disease: proteinuria. Ren Fail. 2013;35(1):62. Epub 2012 Nov 23.
(17:26) Rudnick MR, Goldfarb S, Wexler L, Ludbrook PA, Murphy MJ, Halpern EF, Hill JA, Winniford M, Cohen MB, VanFossen DB. (1995). Nephrotoxicity of ionic and nonionic contrast media in 1196 patients: a randomized trial. The Iohexol Cooperative Study. Kidney Int.;47(1):254.
(21:30) Nijssen EC, Rennenberg RJ, Nelemans PJ, Essers BA, Janssen MM, Vermeeren MA, Ommen VV, Wildberger JE. (2017). Prophylactic hydration to protect renal function from intravascular iodinated contrast material in patients at high risk of contrast-induced nephropathy (AMACING): a prospective, randomised, phase 3, controlled, open-label, non-inferiority trial. Lancet; 389(10076):1312. Epub 2017 Feb 21.
(22:03) Brar, Somjot S., et al. “Haemodynamic-guided fluid administration for the prevention of contrast-induced acute kidney injury: the POSEIDON randomised controlled trial.” The Lancet 383.9931 (2014): 1814-1823.
(23:16) Weisbord SD, Gallagher M, Jneid H, Garcia S, Cass A, Thwin SS, Conner TA, Chertow GM, Bhatt DL, Shunk K, Parikh CR, McFalls EO, Brophy M, Ferguson R, Wu H, Androsenko M, Myles J, Kaufman J, Palevsky PM. Outcomes after Angiography with Sodium Bicarbonate and Acetylcysteine. PRESERVE Trial Group N Engl J Med. 2018;378(7):603. Epub 2017 Nov 12.
(25:06) Shemin, Douglas, et al. “Residual renal function and mortality risk in hemodialysis patients.” American journal of kidney diseases 38.1 (2001): 85-90.
(25:47) ACR Manual on Contrast Media, Version 10.3. (2017). ACR Committee on Drugs and Contrast Media.
