 By Daniel Martinez-Krams
By Daniel Martinez-Krams
Peer ReviewedÂ
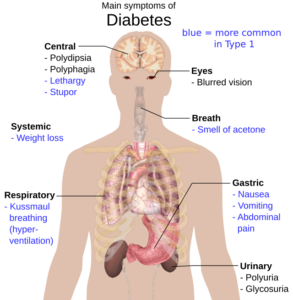
You are rounding on your patients, and your team is discussing a 52-year-old man with HIV who came in with shortness of breath and fever and was found to …

 By Daniel Martinez-Krams
By Daniel Martinez-Krams
Peer ReviewedÂ
You are rounding on your patients, and your team is discussing a 52-year-old man with HIV who came in with shortness of breath and fever and was found to …
 By Daniel Joyce
By Daniel Joyce
Peer Reviewed
I vividly recall my first Impossible Whopper at Burger King after its introduction to the menu in 2019. As I bit down, …
 By Johain Ounadjela
By Johain Ounadjela
Peer Reviewed
I still remember filling out the application form for my driver’s license at the Redwood City, California Department of Motor Vehicles. I was 16 years old and ready …

By Dr. Michael Tanner
The author, at age 14 in 1968, with pennies on his eyes, wearing a Napoleonic military jacket fashionable at the time.
Albums clockwise from top left:
After Bathing at Baxter’s (Jefferson Airplane),
Blonde on Blonde (Bob Dylan),
Disraeli Gears (Cream),
Magical Mystery …
 By Carolyn Akers
By Carolyn Akers
Peer Reviewed
Roger Daltrey, co-founder and lead singer of The Who, had this to say of bandmate Pete Townshend’s famous penchant for bludgeoning Fender Stratocasters:
“Pete wasn’t just smashing his guitar. …
 By Alison Cline
By Alison Cline
Peer Reviewed
Let’s Start with a Case…
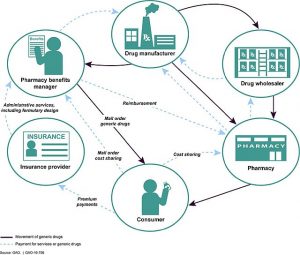
A 79-year-old man with a history of atrial fibrillation and stroke presents to the emergency department with new focal neurologic deficits in the setting of …
 Â By Eric Hu
 By Eric Hu
Peer Reviewed
“May I have the patient’s first name, last name, and medical record number?†The fellow, shooting a piercing glance at the interpreter through the wall-mounted phone, broadcasted her …
 By Olivia Descorbeth
By Olivia Descorbeth
Peer Reviewed
As individuals advance in age, they tend to accumulate medical conditions that require a bevy of pharmaceutical treatments to manage. As a result, polypharmacy, generally defined as the …