 By Robert Mazgaj
By Robert Mazgaj
Faculty Peer Reviewed
Morgellons disease is an “unexplained dermopathy†characterized by fibers emerging from skin lesions, and associated with various cutaneous sensations.[1] Inspired by a curious medical condition reported by …

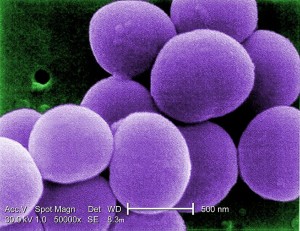 By Bryan Stierman
By Bryan Stierman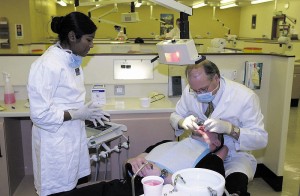 By Jeffrey Krutoy, DDS
By Jeffrey Krutoy, DDS By Nicole Sunseri
By Nicole Sunseri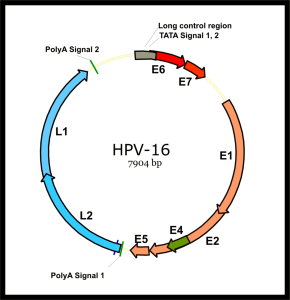 By Kevin Burns
By Kevin Burns By Santosh Vardhana, MD/PhD
By Santosh Vardhana, MD/PhD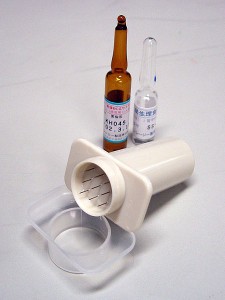 By Mitchell Kim
By Mitchell Kim Please enjoy this post from the Archives, first published on July 30, 2009
Please enjoy this post from the Archives, first published on July 30, 2009