 By Lee Brake
By Lee Brake
Peer Reviewed
Kratom (pronounced KRAY-tum) is a product derived from the crushed and dried leaves of the Mitragyna speciosa plant species endemic to Southeast Asia.1 It has been increasingly used in …

 By Lee Brake
By Lee Brake
Peer Reviewed
Kratom (pronounced KRAY-tum) is a product derived from the crushed and dried leaves of the Mitragyna speciosa plant species endemic to Southeast Asia.1 It has been increasingly used in …
 By Zoe Lawrence, MD
By Zoe Lawrence, MD
Peer Reviewed
The patient is a 43-year-old intravenous drug user who presents to the emergency department with a few days of fevers and malaise. On exam, he is febrile …
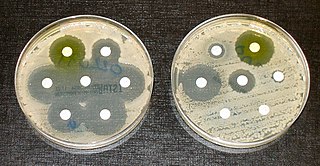 By Avani Kolla
By Avani Kolla
Peer Reviewed
During my trip to India, the “family bonding†reached a new level when I shared my upper respiratory infection with my parents and sister. On day two of …
 By Nina Shevzov-Zebrun
By Nina Shevzov-Zebrun
Peer Reviewed
“How much would I have to pay you to lick the subway floor?â€
“How much money would it take for you to wear an inflatable T-Rex costume to school?â€
Growing …
 Daniel Gratch, MD
Daniel Gratch, MD
Peer reviewed
Although obstructive sleep apnea (OSA) is a disease as ancient as snoring itself, the condition was not fully characterized until the 19th century [1]. In 1889, British physician …
 By Brianna Knoll, MD
By Brianna Knoll, MD
Peer Reviewed
Achieving universal healthcare is arguably one of the central topics of political debate that our new administration will be facing. While it may seem …
 By Ofer Fass, MD
By Ofer Fass, MD
Peer Reviewed
Introduction
QT interval prolongation is a common side effect shared among the most effective antiemetic classes. The risk of QT prolongation is exacerbated by electrolyte abnormalities …
By Varun Subashchandran
Peer Reviewed
Heart failure is a common health condition that is associated with significant morbidity and mortality. The American Heart Association estimates a prevalence of 6.2 million people in the United States between 2013 and 2016.1 In an international pooled analysis of 1506 patients with chronic heart failure, …