 By Rachel Kaplan Hoffmann, M.D., M.S.Ed., and Keith Hoffmann, J.D.
By Rachel Kaplan Hoffmann, M.D., M.S.Ed., and Keith Hoffmann, J.D.
Peer Reviewed
On December 6, 2013, a two-year-old boy living in southeastern Guinea became the first victim of the …

 By Rachel Kaplan Hoffmann, M.D., M.S.Ed., and Keith Hoffmann, J.D.
By Rachel Kaplan Hoffmann, M.D., M.S.Ed., and Keith Hoffmann, J.D.
Peer Reviewed
On December 6, 2013, a two-year-old boy living in southeastern Guinea became the first victim of the …
 By Nathan King
By Nathan King
Faculty Reviewed
Doctors are known to be some of the worst patients, and from personal experience I predict that medical students are not too far behind. That’s why when I finally found the time to take a proactive step …
 By Luke O’Donnell, MD
By Luke O’Donnell, MD
Peer reviewed
Once formidable diseases, pneumonia, bacteremia, and meningitis are all now considered “bread-and-butter” internal medicine. Streptococcus pneumoniae is one of the …
 By Pritha Subramanyam
By Pritha Subramanyam
Peer Reviewed
Mrs. CS is a 66-year-old Indian female who presents for a cardiology follow-up. The patient has a history of mitral regurgitation secondary to …
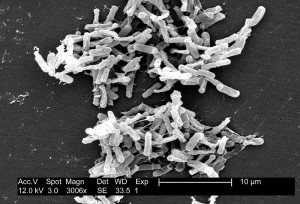 By Theresa Sumberac, MD
By Theresa Sumberac, MD
Peer Reviewed
Antibiotic associated diarrhea is a common complication of antibiotic therapy, occurring in 5% to 39% of all patients receiving treatment. Nearly one third of these cases …
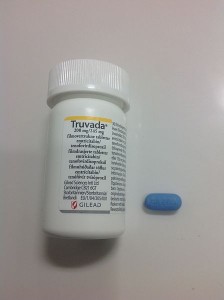 By Richard E. Greene, MD
By Richard E. Greene, MD
Peer Reviewed
In July of 2012, the FDA approved the use of Tenofovir-Emtricitabine (Truvada, a single blue pill) daily as Pre-Exposure Prophylaxis …
 By Aaron Smith, MD
By Aaron Smith, MD
Peer Reviewed
It’s become a familiar site to travelers: airline passengers wearing respiratory masks to filter pathogens from the cabin air. To those not wearing masks, the …
 By Aaron Smith, MD
By Aaron Smith, MD
Peer Reviewed
First introduced in the late 1980s, proton pump inhibitors (PPIs) have revolutionized the treatment of gastric acid-related disorders and have been described as a miracle drug …